Chapter 6
Electronic Structure and Periodic Properties of Elements
Shaun Williams, PhD
Electromagnetic Radiation
- Different Colored Fireworks
Questions to Consider
- Why do we get colors?
- Why do different chemicals give us different colors?

Electromagnetic Radiation
- One of the ways that energy travels through space.
- Three characteristics:
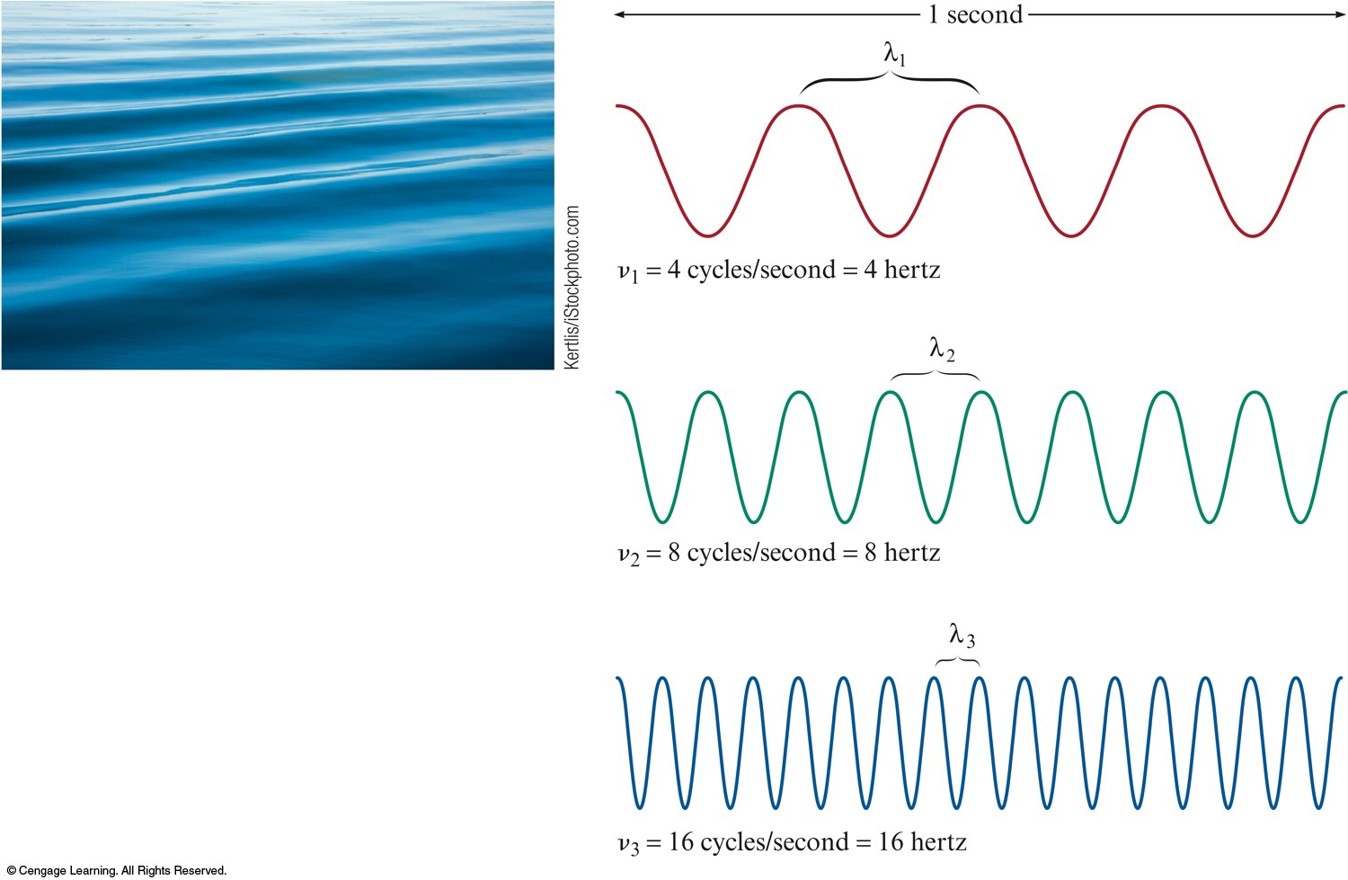
- Wavelength (\(\lambda\)) - distance between two consecutive peaks or troughs in a wave.
- Frequency (\(\nu\)) - number of waves (cycles) per second that pass a given point in space
- Speed (\(c\)) - speed of light (\(2.9979\times 10^8\,\bfrac{\text{m}}{\text{s}}\))
Waves
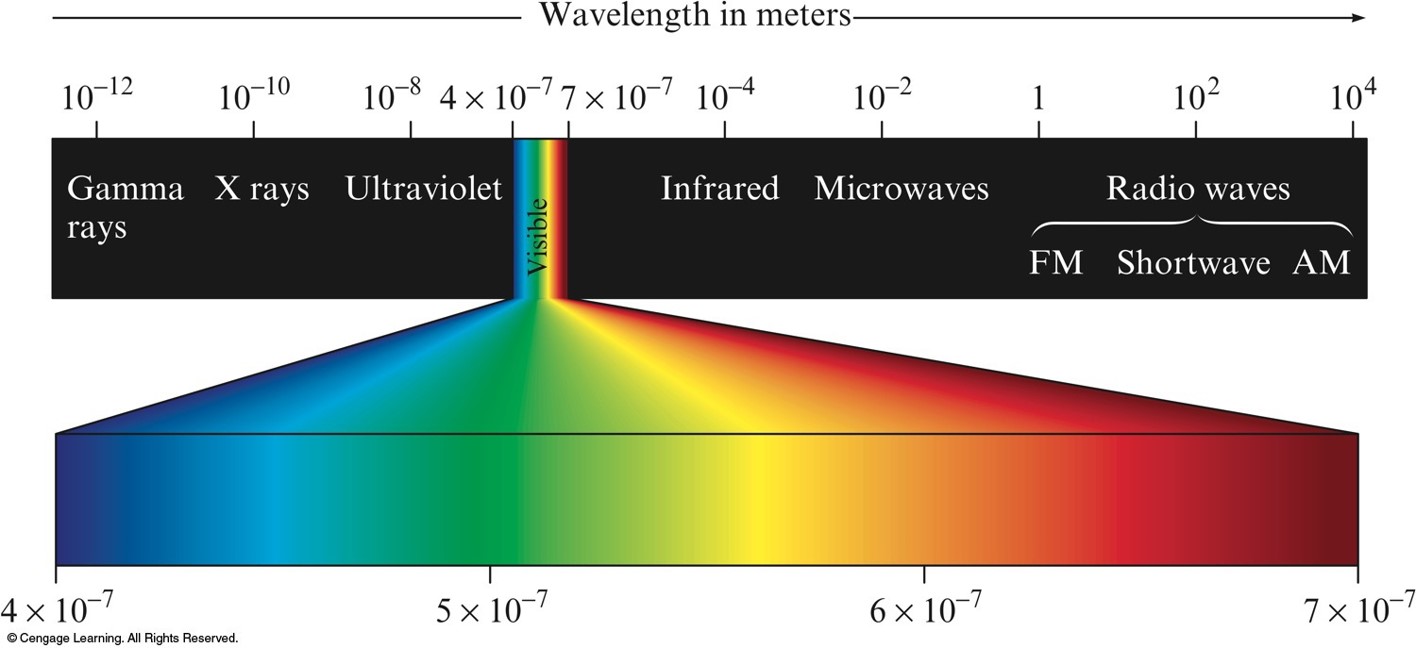
Classification of Electromagnetic Radiation
Pickle Light

The Nature of Matter
- Energy can be gained or lost only in whole number multiples of \(h\nu\).
- A system can transfer energy only in whole quanta (or "packets").
- Energy seems to have particulate properties too.
- Energy is quantized.
- Electromagnetic radiation is a stream of "particles" called photons. $$ E_\text{photon} = h\nu = \frac{hc}{\lambda} $$
- Planck's constant = \(h = 6.626 \times 10^{-34}\,\chem{J\cdot s}\)
Energy
- Energy has mass \(E=mc^2\)
- Dual nature of light:
- Electromagnetic radiation (and all matter) exhibits wave properties and particulate properties.
The Atomic Spectrum of Hydrogen
- Continuous spectrum (results when white light is passed through a prism) - contains all the wavelengths of visible light
- Line spectrum - each line corresponds to a discrete wavelength:
- Hydrogen emission spectrum
Significance
- Only certain energies are allowed for the electron in the hydrogen atom.
- Energy of the electron in the hydrogen atom is quantized.
The Bohr Model
- Electron in a hydrogen atom moves around the nucleus only in certain allowed circular orbits.
- Bohr's model gave hydrogen atom energy levels consistent with the hydrogen emission spectrum.
- Ground state - lowest possible energy state (\(n = 1\))
Electronic Transitions in the Bohr Model for the Hydrogen Atom
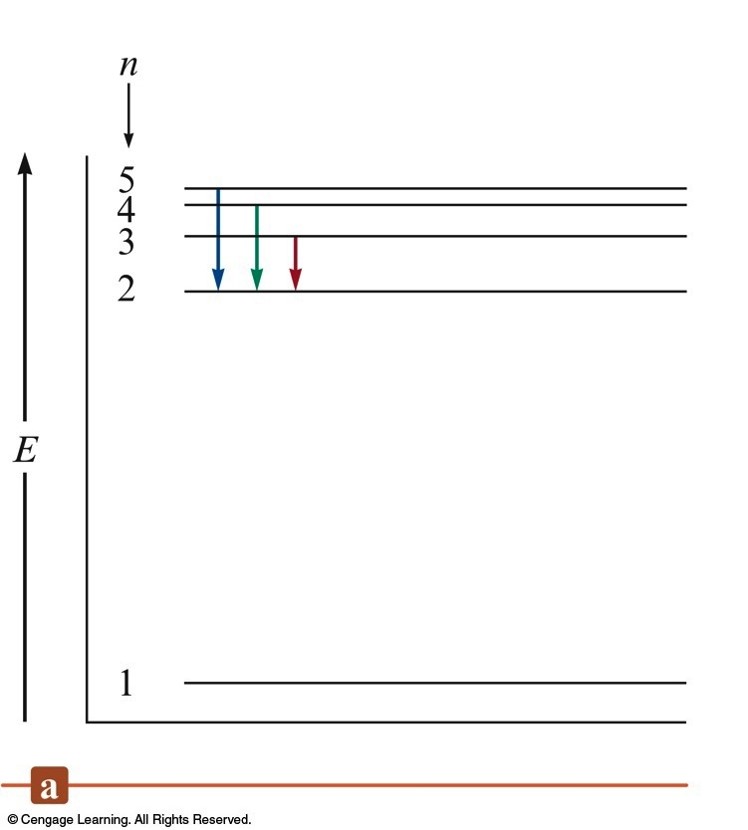
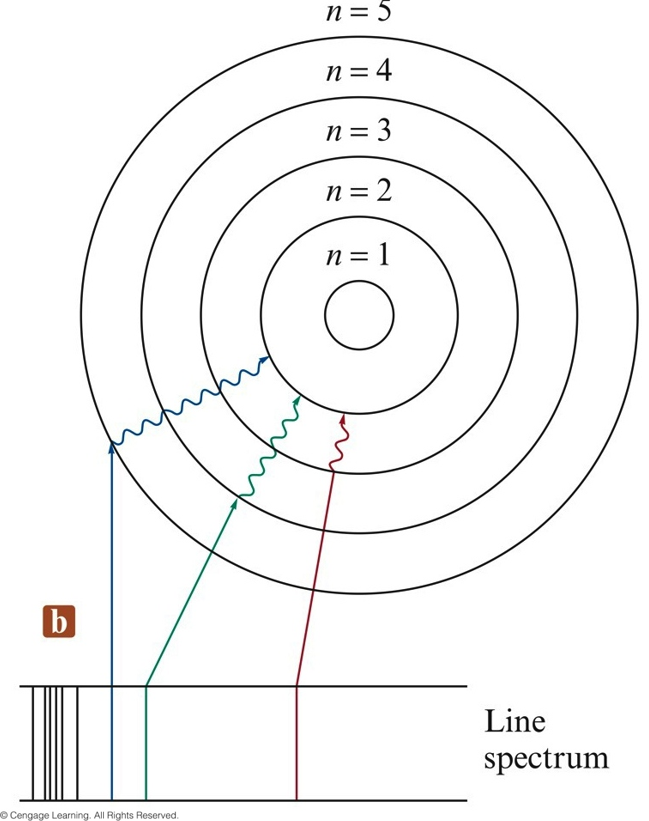
The Bohr Model Energy Equation
- For a single electron transition from one energy level to another:
$$ \Delta E = -2.178 \times 10^{-18}\,\chem{J} \left( \frac{1}{n_\text{final}^2} - \frac{1}{n_\text{initial}^2} \right) $$
- \(\Delta E\) = change in energy of the atom (energy of the emitted photon)
- \(n_\text{final}\) = integer; final distance from the nucleus
- \(n_\text{initial}\) = integer; initial distance from the nucleus
The Bohr Model Analysis
- The model correctly fits the quantized energy levels of the hydrogen atom and postulates only certain allowed circular orbits for the electron.
- As the electron becomes more tightly bound, its energy becomes more negative relative to the zero-energy reference state (free electron). As the electron is brought closer to the nucleus, energy is released from the system.
- Bohr's model is incorrect. This model only works for hydrogen.
- Electrons move around the nucleus in circular orbits.
The Quantum Mechanical Model of the Atom
- We do not know the detailed pathway of an electron.
- Heisenberg uncertainty principle:
- There is a fundamental limitation to just how precisely we can know both the position and momentum of a particle at a given time.
$$ \Delta x \cdot \Delta \left( mv \right) \ge \frac{h}{2\pi} $$
- \(\Delta x\) = uncertainty in a particle's position
- \(\Delta (mν)\) = uncertainty in a particle's momentum
- \(h\) = Planck's constant
- There is a fundamental limitation to just how precisely we can know both the position and momentum of a particle at a given time.
$$ \Delta x \cdot \Delta \left( mv \right) \ge \frac{h}{2\pi} $$
Physical Meaning of a Wave Function \((\Psi)\)
- The square of the function indicates the probability of finding an electron near a particular point in space.
- Probability distribution – intensity of color is used to indicate the probability value near a given point in space.
Probability Distribution for the 1s Wave Function
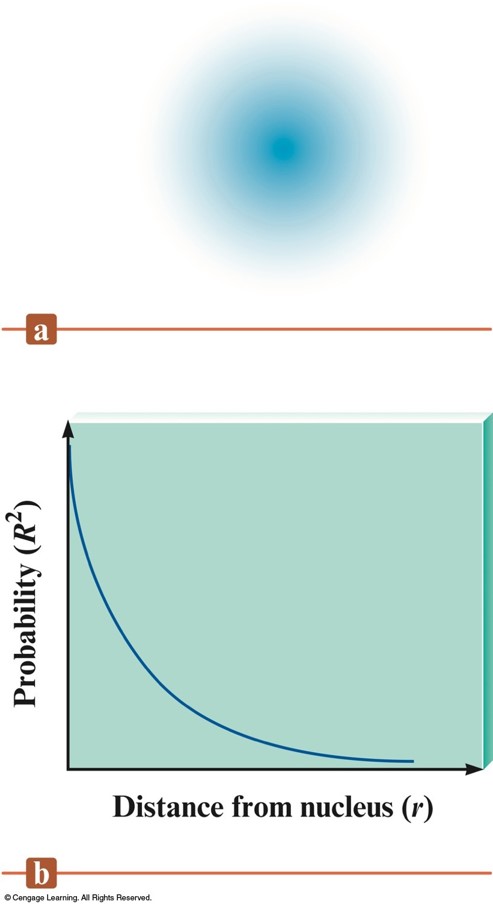
Radial Probability Distribution
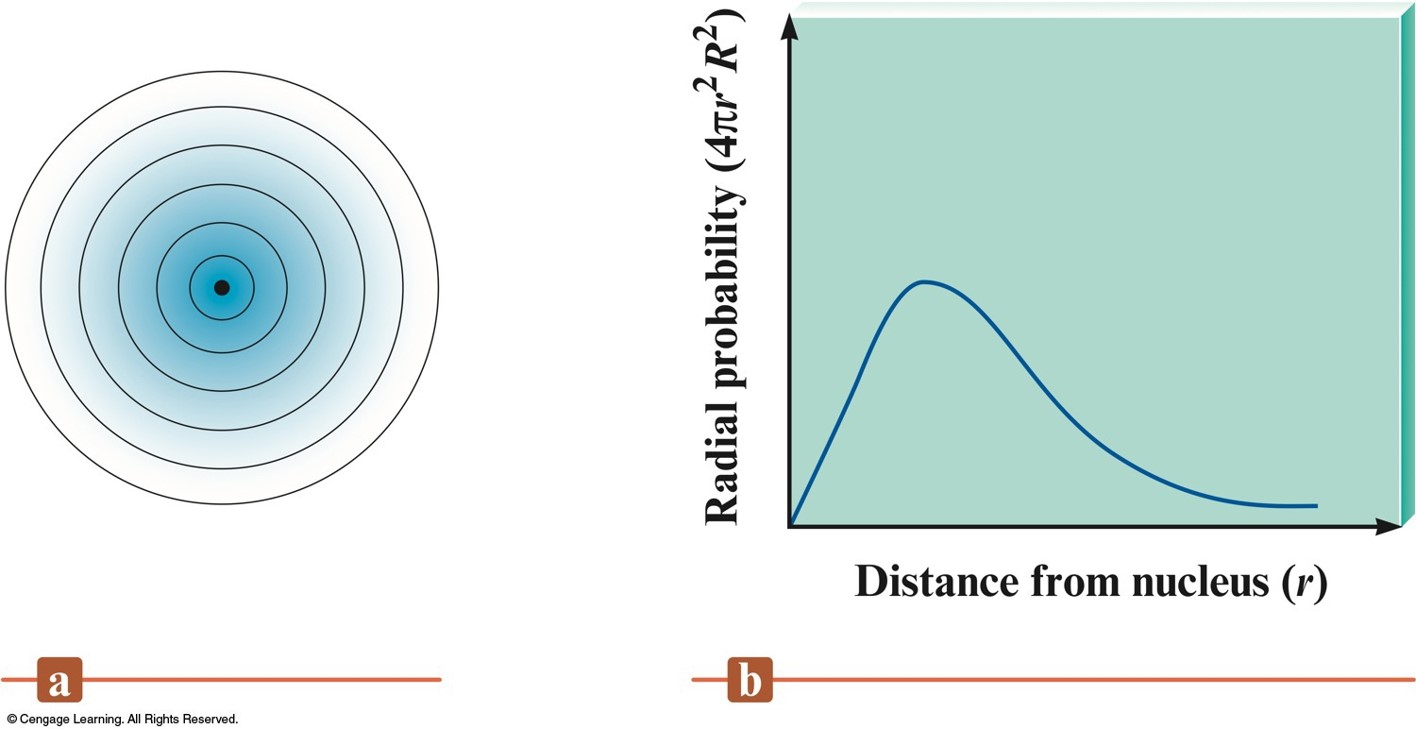
Relative Orbital Size
- Difficult to define precisely.
- Orbital is a wave function.
- Picture an orbital as a three-dimensional electron density map.
- Hydrogen 1s orbital:
- Radius of the sphere that encloses 90% of the total electron probability.
Quantum Numbers
- Principal quantum number (\(n\)) – size and energy of the orbital.
- Angular momentum quantum number (\(l\)) – shape of atomic orbitals (sometimes called a subshell).
- Magnetic quantum number (\(m_l\)) – orientation of the orbital in space relative to the other orbitals in the atom.
Quantum Numbers for the First Four Levels of Orbitals in the Hydrogen Atom
| \(n\) | \(l\) | Sublevel Designation |
\(m_l\) | Number of Orbitals |
|||
|---|---|---|---|---|---|---|---|
| 1 | 0 | \(1s\) | 0 | 1 | |||
| 2 | 0 | \(2s\) | 0 | 1 | |||
| 1 | \(2p\) | -1,0,+1 | 3 | ||||
| 3 | 0 | \(3s\) | 0 | 1 | |||
| 1 | \(3p\) | -1,0,+1 | 3 | ||||
| 2 | \(3d\) | -2,-1,0,+1,+2 | 5 | ||||
| 4 | 0 | \(4s\) | 0 | 1 | |||
| 1 | \(4p\) | -1,0,+1 | 3 | ||||
| 2 | \(4d\) | -2,-1,0,+1,+2 | 5 | ||||
| 3 | \(4f\) | -3,-2,-1,0,+1,+2,+3 | 7 |
Exercise 1
For principal quantum level \(n = 3\), determine the number of allowed subshells (different values of \(l\)), and give the designation of each.
Exercise 1 - Answer
For principal quantum level \(n = 3\), determine the number of allowed subshells (different values of \(l\)), and give the designation of each.
Number of allowed subshells = 3 (\(l=0,1,2\))
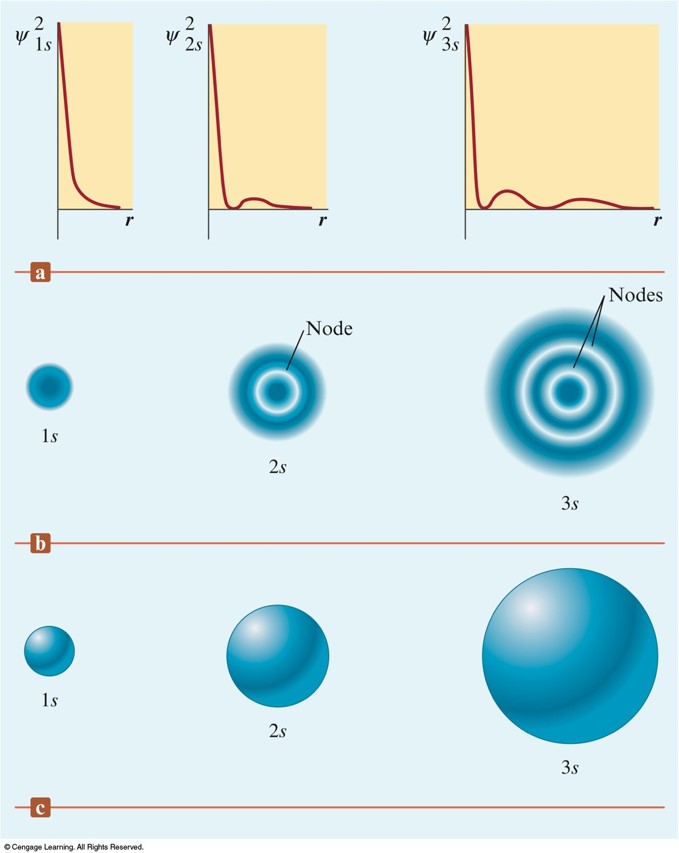
Orbital Shapes and Energies
Three Representations of the Hydrogen \(1s\), \(2s\), and \(3s\) Orbitals
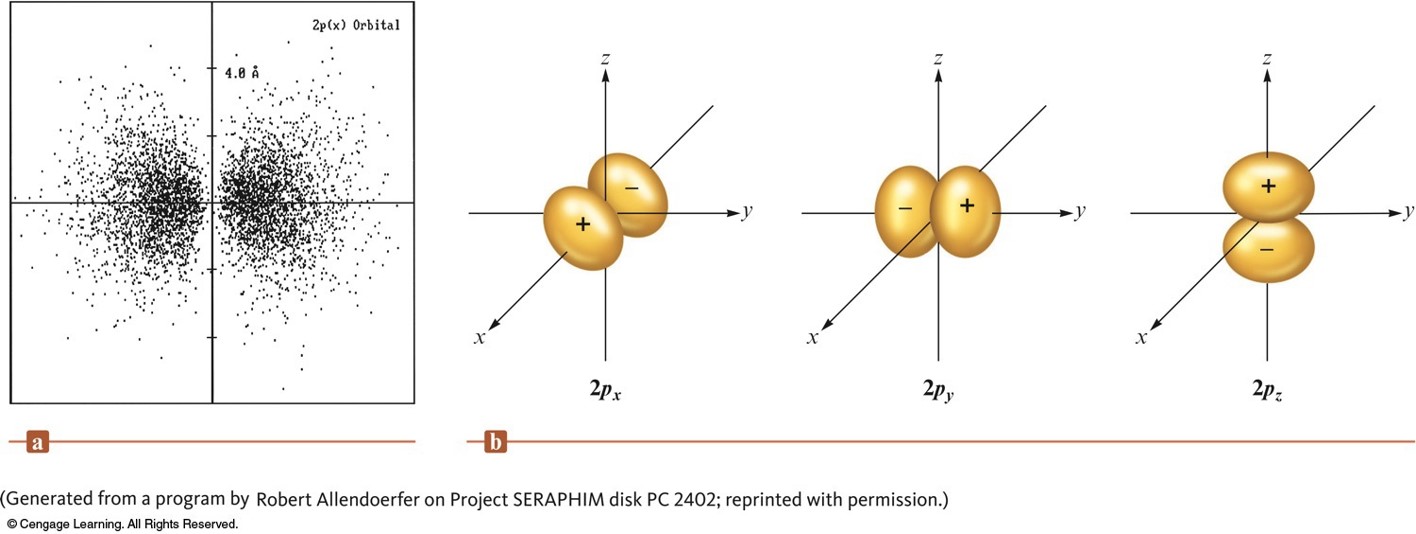
The Boundary Surface Representations of All Three \(2p\) Orbitals
The Boundary Surfaces of All of the \(3d\) Orbitals
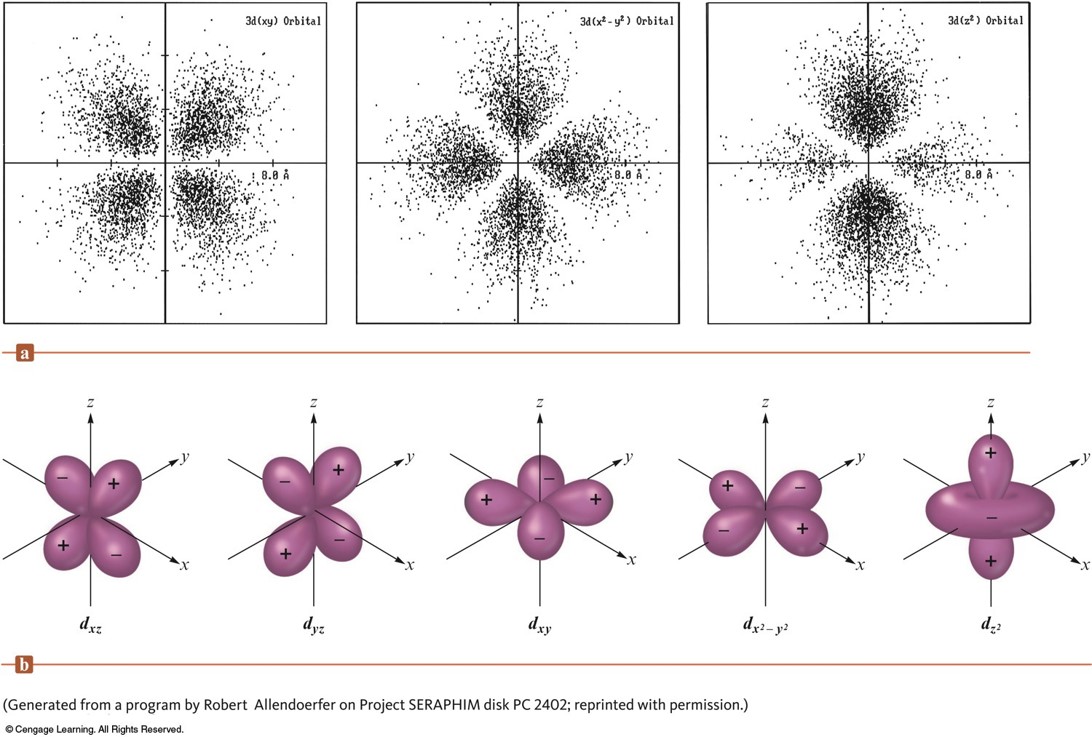
Representation of the \(4f\) Orbitals in Terms of Their Boundary Surfaces
Electron Spin and the Pauli Principle
Electron Spin
- Electron spin quantum number (\(m_s\)) - can be \(+\bfrac{1}{2}\) or \(-\bfrac{1}{2}\).
- Pauli exclusion principle - in a given atom no two electrons can have the same set of four quantum numbers.
- An orbital can hold only two electrons, and they must have opposite spins.
Polyelectronic Atoms
- Atoms with more than one electron.
- Electron correlation problem:
- Since the electron pathways are unknown, the electron repulsions cannot be calculated exactly.
- When electrons are placed in a particular quantum level, they "prefer" the orbitals in the order \(s\), \(p\), \(d\), and then \(f\).
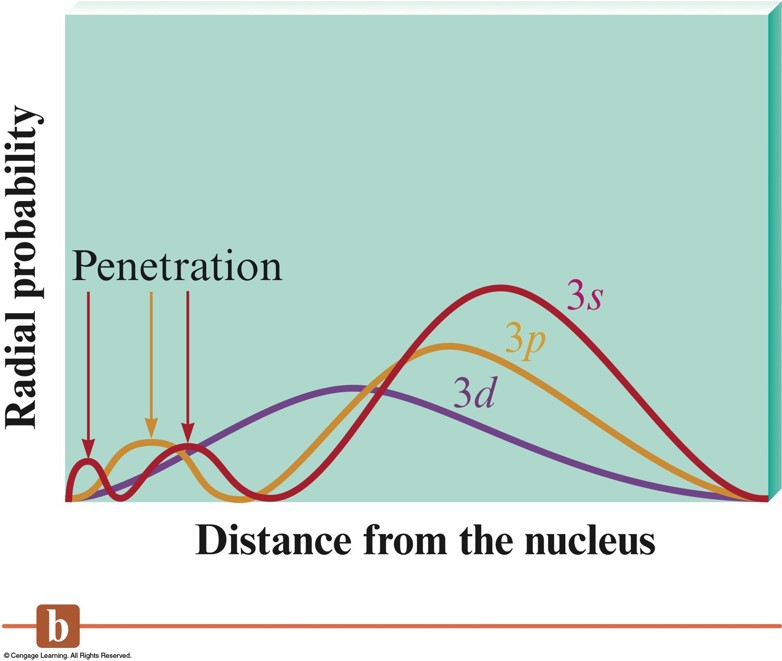
Penetration Effect
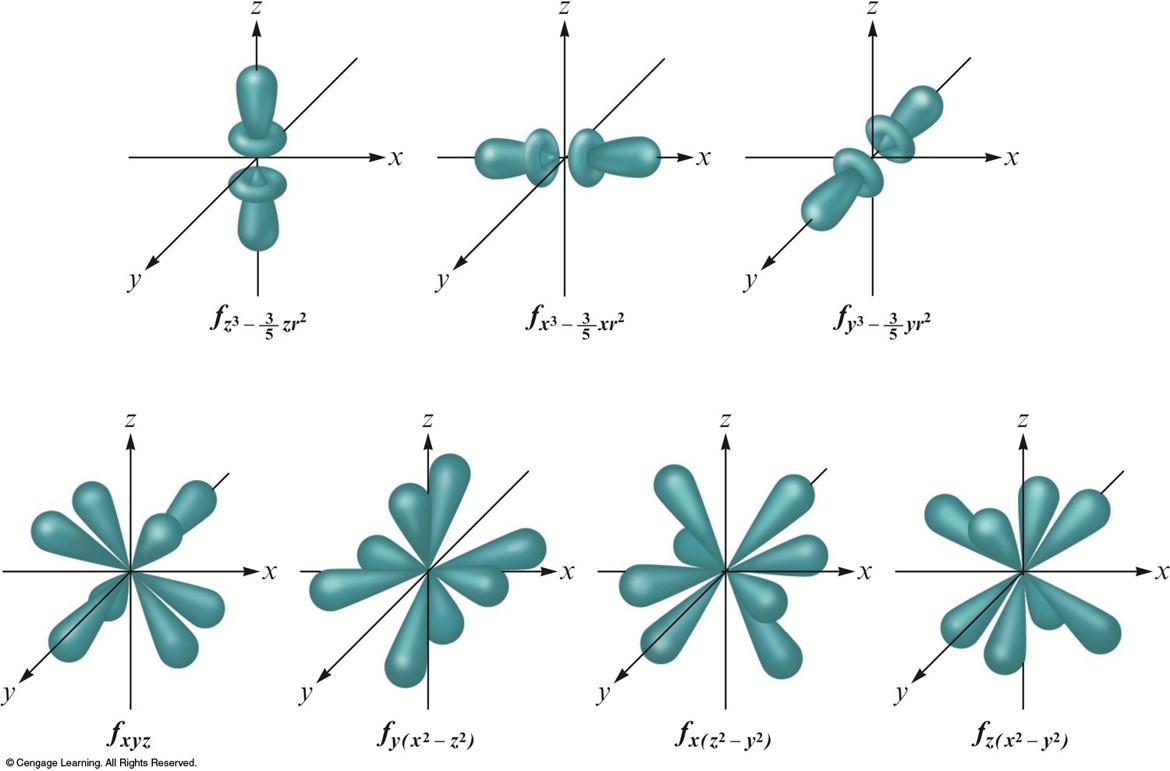
- A \(2s\) electron penetrates to the nucleus more than one in the \(2p\) orbital.
- This causes an electron in a \(2s\) orbital to be attracted to the nucleus more strongly than an electron in a \(2p\) orbital.
- Thus, the \(2s\) orbital is lower in energy than the \(2p\) orbitals in a polyelectronic atom.
A Comparison of the Radial Probability Distributions of the \(2s\) and \(2p\) Orbitals
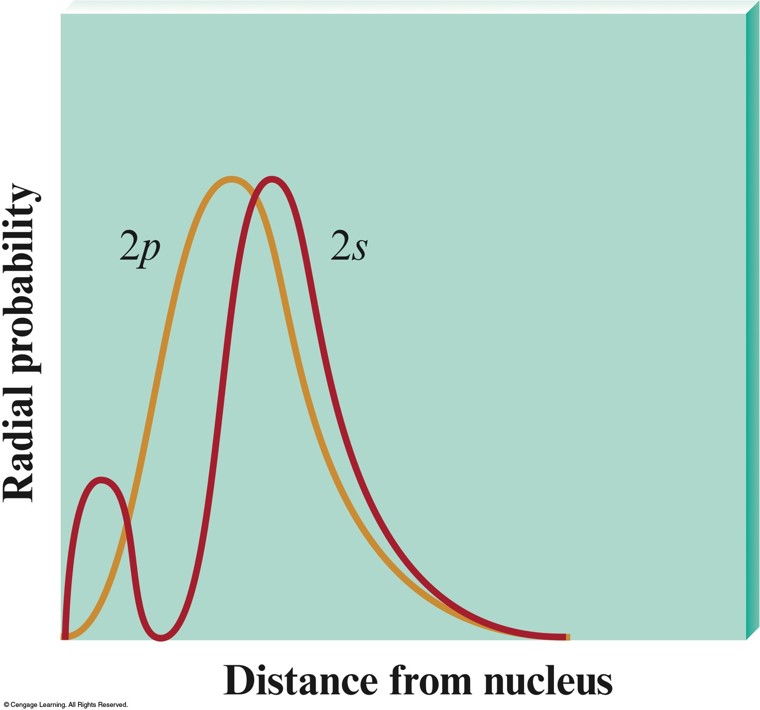
The Radial Probability Distribution of the \(3s\) Orbital
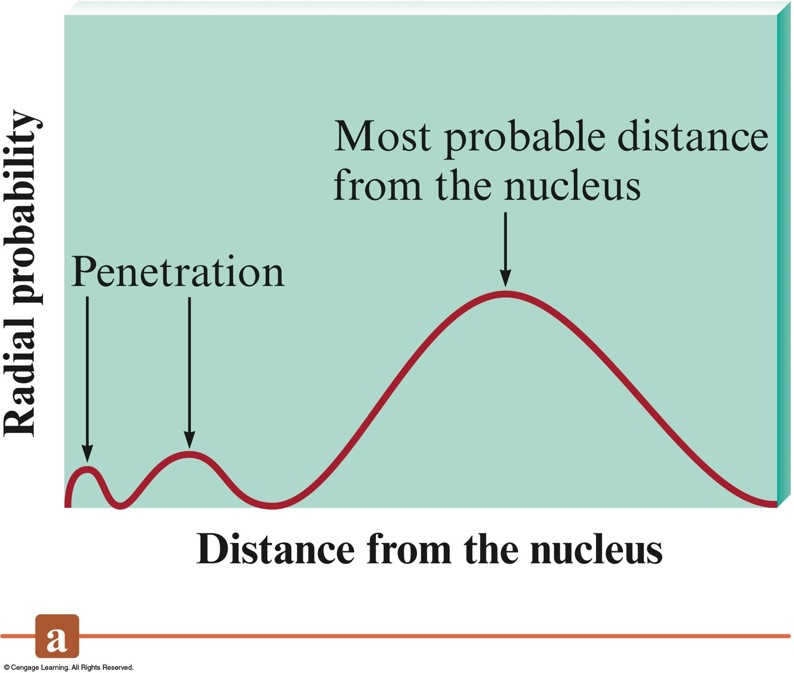
A Comparison of the Radial Probability Distributions of the \(3s\), \(3p\), and \(3d\) Orbitals
The History of the Periodic Table
- Originally constructed to represent the patterns observed in the chemical properties of the elements.
- Mendeleev is given the most credit for the current version of the periodic table because he emphasized how useful the periodic table could be in predicting the existence and properties of still unknown elements.
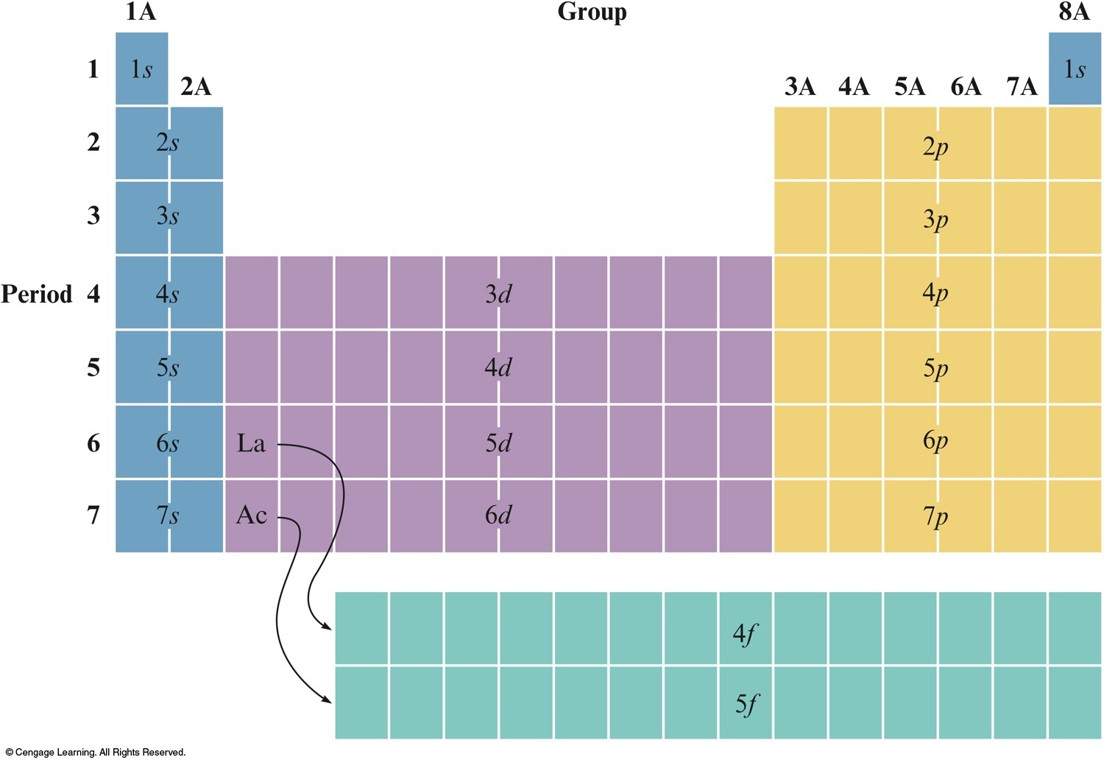
The Aufbau Principle and the Periodic Table
Aufbau Principle
- As protons are added one by one to the nucleus to build up the elements, electrons are similarly added to hydrogen-like orbitals.
- An oxygen atom has an electron arrangement of two electrons in the \(1s\) subshell, two electrons in the \(2s\) subshell, and four electrons in the \(2p\) subshell.
Oxygen: \(1s^22s^22p^4\)
Hund's Rule
- The lowest energy configuration for an atom is the one having the maximum number of unpaired electrons allowed by the Pauli principle in a particular set of degenerate (same energy) orbitals.
Orbital Diagram
- A notation that shows how many electrons an atom has in each of its occupied electron orbitals.
Oxygen: \(1s^22s^22p^4\)
| \(1s\) | \(\phantom{2s}\) | \(2s\) | \(\phantom{2s}\) | \(2p\) | ||
|---|---|---|---|---|---|---|
| \(\uparrow\downarrow\) | \(\phantom{\uparrow\downarrow}\) | \(\uparrow\downarrow\) | \(\phantom{\uparrow\downarrow}\) | \(\uparrow\downarrow\) | \(\uparrow\phantom{\downarrow}\) | \(\uparrow\phantom{\downarrow}\) |
Valence Electrons
- The electrons in the outermost principal quantum level of an atom. \(1s^22s^22p^6\) (valence electrons = 8)
- The elements in the same group on the periodic table have the same valence electron configuration.
The Orbitals Being Filled for Elements in Various Parts of the Periodic Table
Exercise 2
Determine the expected electron configurations for each of the following.
- S
- Ba
- Eu
Exercise 2 - Answer
Determine the expected electron configurations for each of the following.
- S - \(1s^22s^22p^63s^23p^4\) or \([\chem{Ne}]3s^23p^4\)
- Ba - \([\chem{Xe}]6s^2\)
- Eu - \([\chem{Xe}]6s^24f^7\)
Periodic Trends in Atomic Properties
Periodic Trends
- Ionization Energy
- Electron Affinity
- Atomic Radius
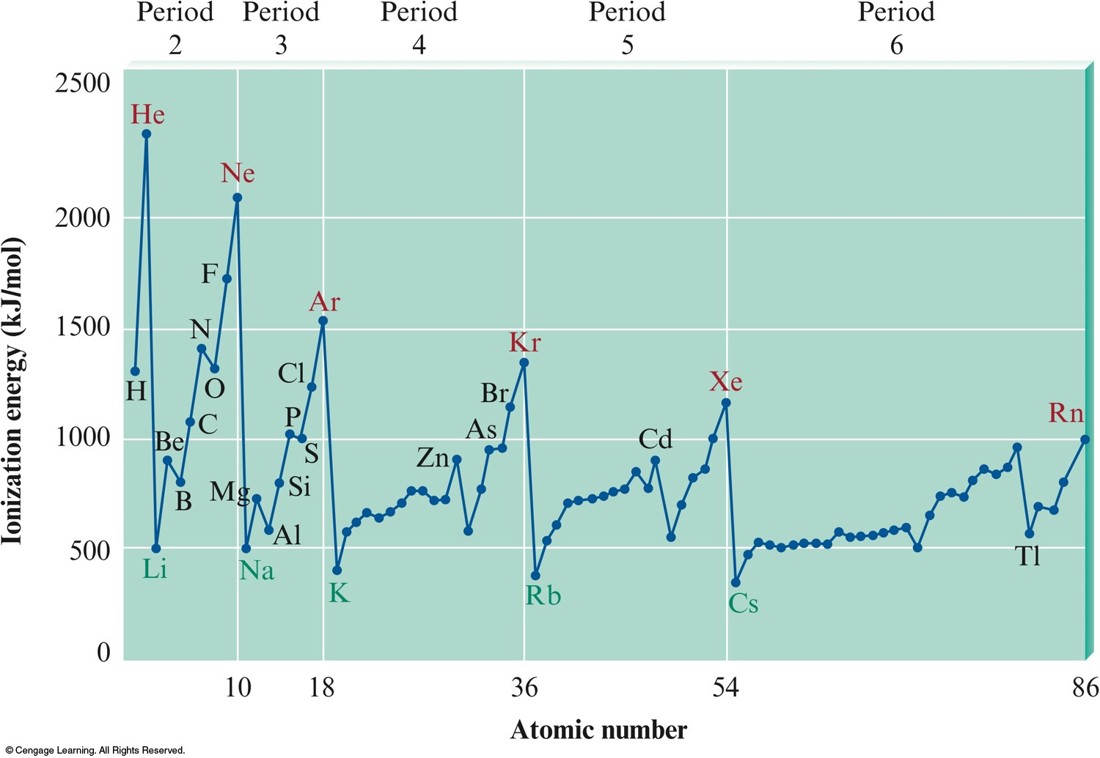
Ionization Energy
- Energy required to remove an electron from a gaseous atom or ion.
- \(X(g)\rightarrow X^+(g)+e^-\)
| \(\chem{Mg\rightarrow Mg^+ + e^-}\) | \(I_1=735\,\bfrac{\chem{kJ}}{\chem{mol}}\) | (1st IE) |
| \(\chem{Mg^+\rightarrow Mg^{2+} + e^-}\) | \(I_2=1445\,\bfrac{\chem{kJ}}{\chem{mol}}\) | (2nd IE) |
| \(\chem{Mg^{2+}\rightarrow Mg^{3+} + e^-}\) | \(I_3=7730\,\bfrac{\chem{kJ}}{\chem{mol}}\) | (3rd IE)* |
* Core electrons are bound much more tightly than valence electrons.
More on Ionization Energy
- In general, as we go across a period from left to right, the first ionization energy increases.
- Why?
- Electrons added in the same principal quantum level do not completely shield the increasing nuclear charge caused by the added protons.
- Electrons in the same principal quantum level are generally more strongly bound from left to right on the periodic table.
- In general, as we go down a group from top to bottom, the first ionization energy decreases.
- Why?
- The electrons being removed are, on average, farther from the nucleus.
The Values of First Ionization Energy for the Elements in the First Six Periods
Successive Ionization Energies (kJ per Mole) for the Elements in Period 3
| Element | \(I_1\) | \(I_2\) | \(I_3\) | \(I_4\) | \(I_5\) | \(I_6\) | \(I_7\) |
|---|---|---|---|---|---|---|---|
| Na | 495 | 4560 | |||||
| Mg | 735 | 1445 | 7730 | ||||
| Al | 580 | 1815 | 2740 | 11600 | |||
| Si | 780 | 1575 | 3220 | 4350 | 16100 | ||
| P | 1060 | 1890 | 2905 | 4950 | 6270 | 21200 | |
| S | 1005 | 2260 | 3375 | 4565 | 6950 | 8490 | 27000 |
| Cl | 1255 | 2295 | 3850 | 5160 | 6560 | 9360 | 11000 |
| Ar | 1527 | 2665 | 3945 | 5770 | 7230 | 8780 | 12000 |
Electron Affinity
- Energy change associated with the addition of an electron to a gaseous atom.
- \(\chem{X(g) + e^– \rightarrow X^–(g)}\)
- In general as we go across a period from left to right, the electron affinities become more negative.
- In general electron affinity becomes more positive in going down a group.
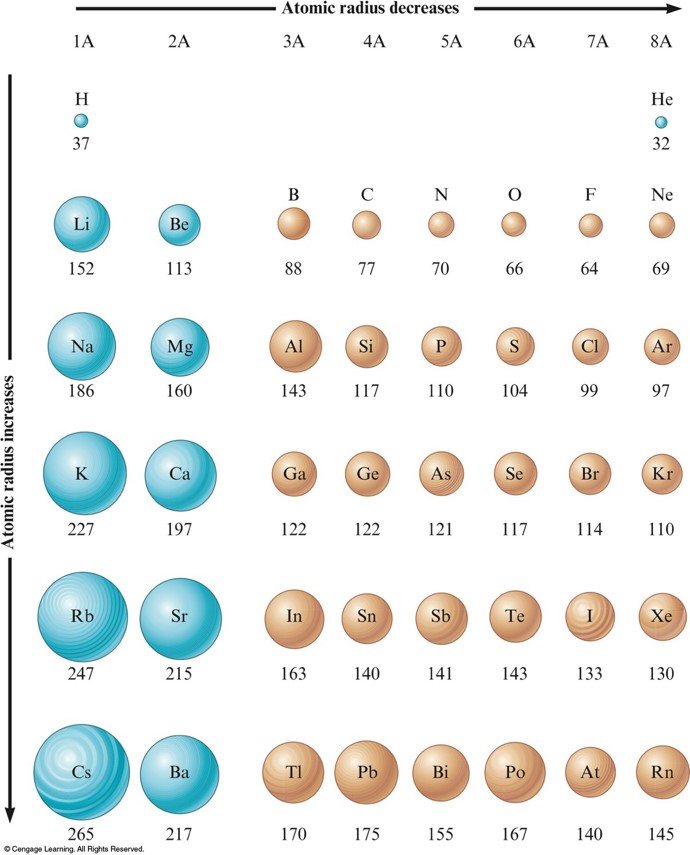
Atomic Radius
- In general as we go across a period from left to right, the atomic radius decreases.
- Effective nuclear charge increases, therefore the valence electrons are drawn closer to the nucleus, decreasing the size of the atom.
- In general atomic radius increases in going down a group.
- Orbital sizes increase in successive principal quantum levels.
Atomic Radii for Selected Atoms
Exercise 3
Arrange the elements oxygen, fluorine, and sulfur according to increasing:
- Ionization energy
- Atomic size
Exercise 3 - Answer
Arrange the elements oxygen, fluorine, and sulfur according to increasing:
- Ionization energy - \( \chem{S,O,F} \)
- Atomic size - \( \chem{F,O,S} \)
The Properties of a Group: The Alkali Metals
The Periodic Table - Final Thoughts
- It is the number and type of valence electrons that primarily determine an atom's chemistry.
- Electron configurations can be determined from the organization of the periodic table.
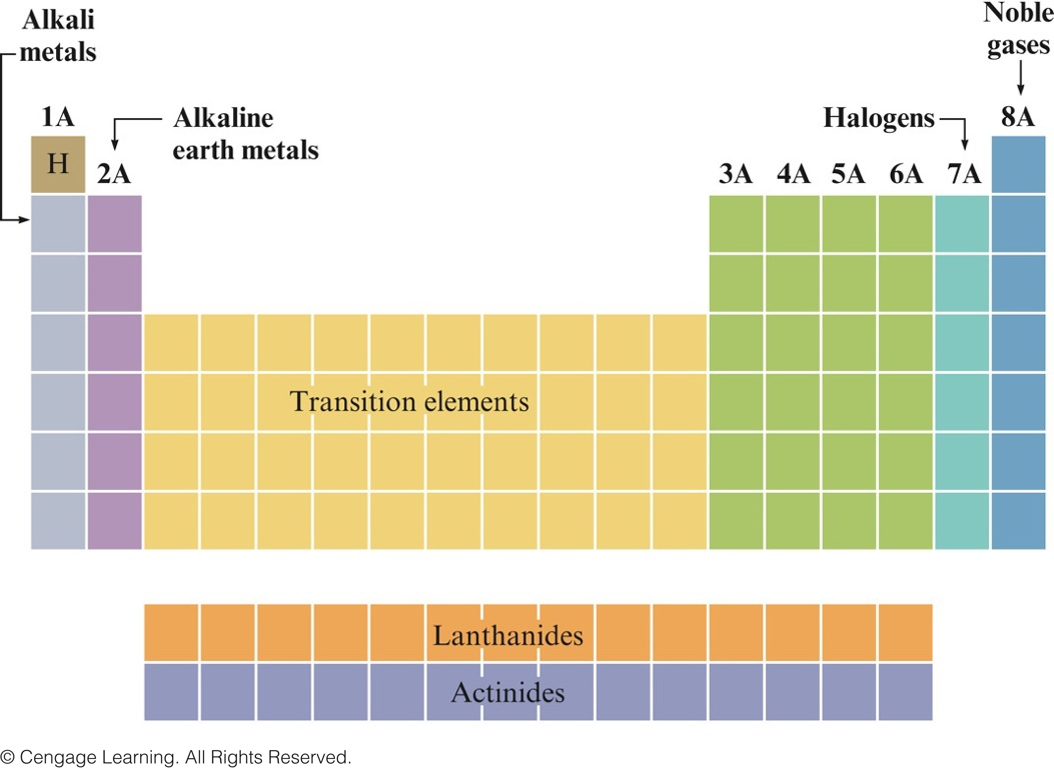
- Certain groups in the periodic table have special names.
Special Names for Groups in the Periodic Table
The Periodic Table - Final Thoughts (cont.)
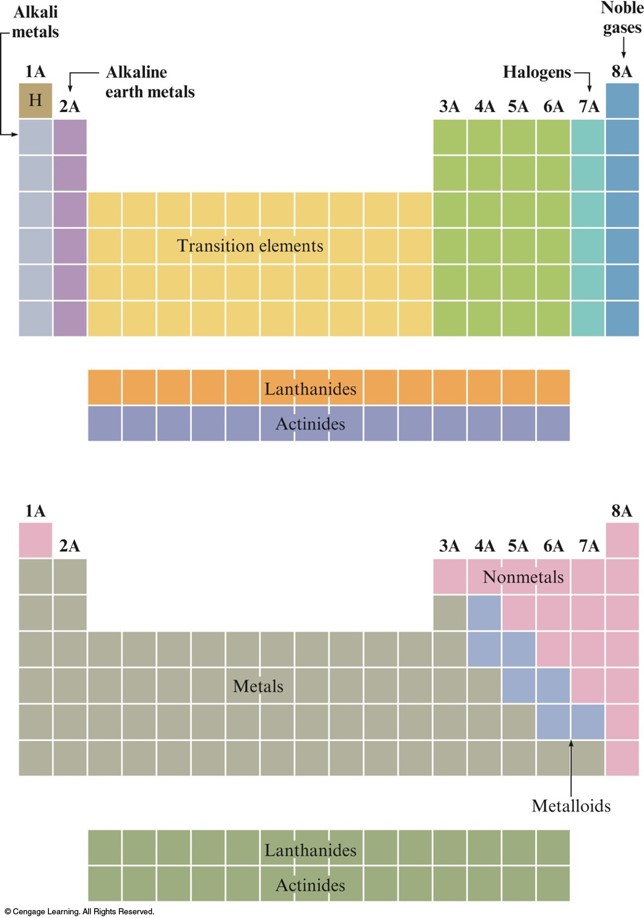
- Basic division of the elements in the periodic table is into metals and nonmetals.
The Alkali Metals
- \(\chem{Li}\), \(\chem{Na}\), \(\chem{K}\), \(\chem{Rb}\), \(\chem{Cs}\), and \(\chem{Fr}\)
- Most chemically reactive of the metals
- React with nonmetals to form ionic solids
- Going down group:
- Ionization energy decreases
- Atomic radius increases
- Density increases
- Melting and boiling points smoothly decrease
- Most chemically reactive of the metals
/